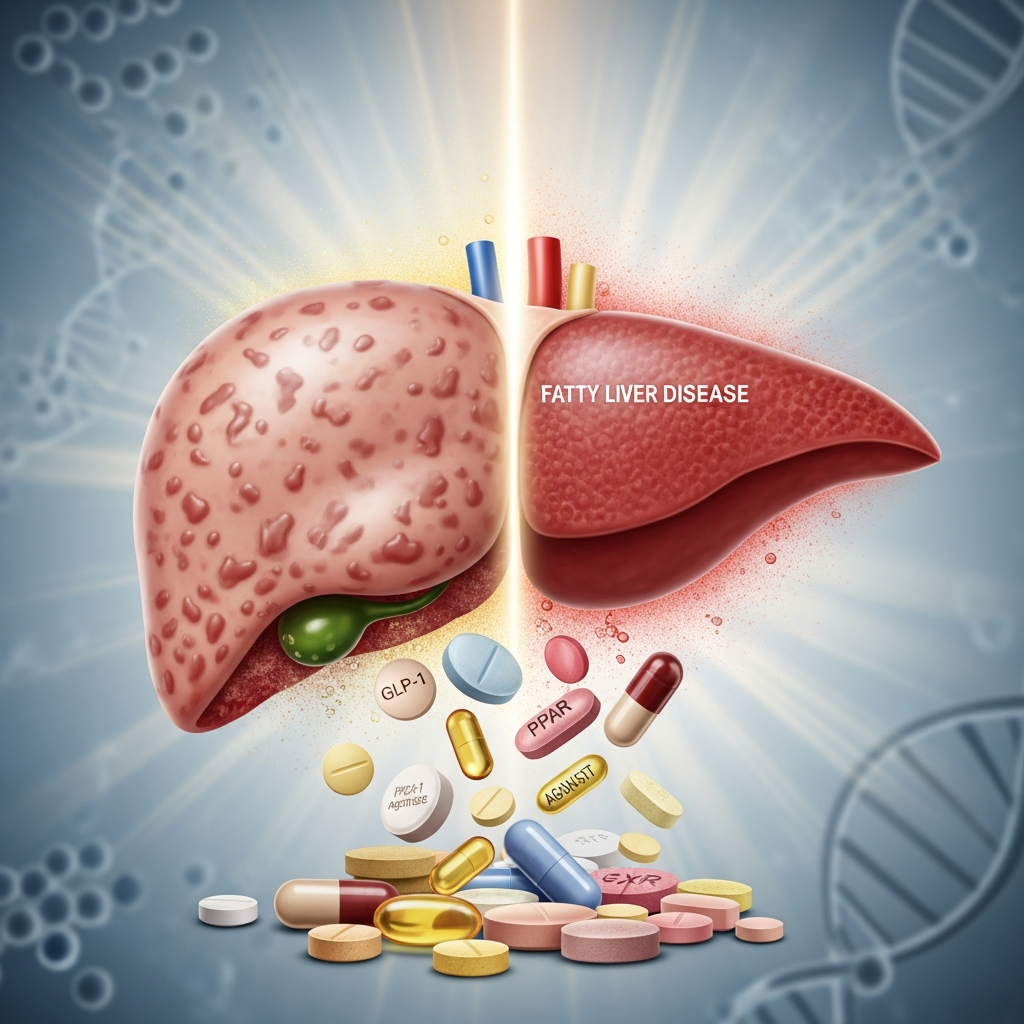
Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD), formerly known as non-alcoholic fatty liver disease (NAFLD), has become the most widespread liver condition globally. It now affects approximately one in three adults. This prevalent disorder occurs when excess fat accumulates within liver cells, potentially leading to severe liver damage. Beyond hepatic complications, MASLD significantly elevates the risk of life-threatening heart and blood vessel diseases. New research offers a compelling ray of hope for millions.
A Global Health Challenge: Understanding MASLD
MASLD is a metabolic disorder closely linked to obesity, insulin resistance, and high blood pressure. Unlike alcoholic fatty liver disease, MASLD develops independently of heavy alcohol consumption. Its progression can range from simple fat accumulation (steatosis) to inflammation (steatohepatitis), fibrosis (scarring), and eventually cirrhosis or even liver cancer. The sheer scale of MASLD presents an immense public health challenge, with current treatment options largely limited to lifestyle modifications, which can be difficult for many patients to sustain. This scarcity of pharmaceutical interventions underscores the critical need for novel therapeutic approaches.
The Promise of Drug Repurposing for Liver Health
For years, scientists have sought effective drug treatments for MASLD. However, many experimental therapies have failed in clinical trials, often due to safety concerns. This has driven researchers to explore drug repurposing—a strategy that involves finding new therapeutic uses for medications already approved for other conditions. This approach offers significant advantages:
Enhanced Safety Profile: Repurposed drugs have established safety records in humans, reducing risks.
Faster Development: They can bypass lengthy early-stage clinical development, accelerating availability.
Cost-Effectiveness: Repurposing is often more economical than developing entirely new compounds.
This strategy is particularly valuable for early-stage MASLD, which often presents without noticeable symptoms. “We have focused on these phases to prevent the disease from progressing,” explains Marta Alegret, a professor at the University of Barcelona’s Faculty of Pharmacy and Food Sciences, who led a significant recent study. “For a drug to be used in these early stages, it must have a good safety profile in humans. That is why we have studied drugs already on the market for other pathologies, which have been shown to be very safe and could have a potential benefit.”
Breakthrough from Barcelona: Pemafibrate and Telmisartan
A recent study from the University of Barcelona, published in Pharmacological Research*, has identified a promising drug combination for MASLD. Researchers discovered that two existing medications, pemafibrate and telmisartan, significantly reduced liver fat in animal models. What’s more, using both drugs together appeared to offer enhanced benefits, not only improving liver health but also reducing associated cardiovascular risks.
Pemafibrate is a lipid-lowering drug currently marketed in Japan, while telmisartan is a widely prescribed medication for high blood pressure globally. Both are already used to manage cardiovascular risk factors. Alegret notes, “Mortality from cardiovascular causes is significant in patients with MASLD, and often these patients also have these two risk factors together.” This dual action against liver fat and cardiovascular risk makes the combination particularly compelling.
How the Combination Therapy Works
The research team tested the drugs in animal models, including rats and zebrafish larvae. Zebrafish are valuable for liver disease studies due to metabolic similarities with humans and their suitability for rapid, affordable experiments. The results were striking: the combination of pemafibrate and telmisartan effectively reversed liver fat buildup caused by diets high in fat and fructose.
A key finding was the synergistic effect: using half doses of both drugs together was as effective as a full dose of either drug alone. Alegret emphasizes, “Combination therapy with drugs acting on different pathogenic pathways may be a better strategy than monotherapy, thanks to possible synergistic effects and reduced toxicity related to the use of lower doses of each drug.” Beyond direct liver benefits, the treatment also showed potential to lower blood pressure and cholesterol levels, further contributing to a reduced cardiovascular risk.
Unveiling a New Mechanism: The Role of PCK1
The study also delved into the distinct biological pathways through which these drugs operate. For the first time, researchers identified a crucial role for the PCK1 protein in telmisartan’s mechanism of action for reducing liver fat. Telmisartan has been studied in MASLD models before, primarily for its anti-inflammatory and anti-fibrotic effects in more advanced stages. However, in early MASLD, where inflammation and fibrosis are not yet present, its beneficial impact on lipid accumulation remained less clear.
In MASLD animal models, PCK1 levels in the liver were lower than normal. Telmisartan treatment successfully restored these levels, shifting how the liver processes nutrients. This increase in PCK1 diverted metabolic flow from lipid synthesis towards glucose synthesis. Crucially, the researchers observed that this shift did not lead to increased glucose levels in the blood, thus avoiding the risk of inducing diabetes. This discovery sheds new light on telmisartan’s therapeutic potential in the early stages of MASLD.
Broader Research Efforts in Liver Disease Treatment
The pursuit of new MASLD treatments is a global endeavor, with several promising avenues being explored, often leveraging the drug repurposing strategy.
Tackling Liver Scarring: Silybin and Carvedilol
While the Barcelona study focused on reversing early-stage fat accumulation, other research is addressing more advanced liver damage. A separate study, for instance, has identified a combination of silybin (derived from milk thistle) and carvedilol (a blood pressure medication) that shows promise in reversing liver scarring, or fibrosis, in laboratory settings and mice. Liver fibrosis, where scar tissue accumulates, can lead to cirrhosis and liver cancer. This research highlights the diverse applications of drug repurposing across different stages of liver disease, demonstrating a comprehensive scientific effort to combat liver conditions.
Plant-Based Hope: Cannabis Compounds for MASLD
Another exciting area of research explores plant-derived compounds. A study from the Hebrew University of Jerusalem suggests that non-intoxicating cannabis compounds, specifically cannabidiol (CBD) and cannabigerol (CBG), could offer a novel approach to treating MASLD. These compounds significantly reduced liver fat and improved metabolic health in experimental models.
The research unveiled a mechanism of “metabolic remodeling,” where CBD and CBG enhance hepatic energy and lysosomal function. They create a “backup battery” for the liver by increasing phosphocreatine, a high-energy molecule, helping the liver function under metabolic stress. Additionally, these compounds restored the activity of cathepsins, enzymes crucial for cellular waste breakdown, enabling more efficient clearance of harmful fats like triglycerides and ceramides. While both CBD and CBG showed benefits, CBG demonstrated more robust improvements in reducing body fat, LDL cholesterol, and improving insulin sensitivity. These studies offer a glimpse into the diverse and innovative strategies being developed to tackle MASLD.
The Road Ahead: From Lab to Clinic
While the findings from the University of Barcelona study are highly promising, the research is still in its early stages. The observed benefits come from animal studies, and rigorous clinical trials are essential to confirm if these effects translate safely and effectively to human patients with MASLD.
Marta Alegret emphasizes, “In order to be translated into a treatment for MASLD patients, clinical studies would be needed to show that the benefits observed in animal models also occur in humans.” The research team is now continuing their work by exploring whether this drug combination could also be effective in more advanced stages of MASLD, particularly when liver fibrosis is present. They are also developing new animal models that include both liver disease and cardiovascular conditions to comprehensively assess if the beneficial actions extend beyond the liver, potentially reducing atherosclerosis and improving overall cardiovascular health.
Frequently Asked Questions
What is Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD)?
MASLD, previously known as NAFLD, is a chronic liver condition affecting about one in three adults globally. It involves excessive fat accumulation in liver cells, which can lead to inflammation, scarring (fibrosis), and serious liver damage like cirrhosis or liver cancer. Beyond liver-specific issues, MASLD significantly increases the risk of cardiovascular diseases, making it a major public health concern closely linked to obesity, insulin resistance, and high blood pressure.
How do pemafibrate and telmisartan work to treat MASLD?
The University of Barcelona study found that pemafibrate, a lipid-lowering drug, and telmisartan, a blood pressure medication, can significantly reduce liver fat. They work synergistically: telmisartan was identified to restore levels of the PCK1 protein in the liver, diverting metabolites away from lipid synthesis towards glucose synthesis without causing high blood sugar. Both drugs are also used to manage cardiovascular risks, offering a dual benefit by improving liver health and reducing associated heart and blood vessel disease risks.
When could these drug treatments for fatty liver disease be available to patients?
While the research on pemafibrate and telmisartan for MASLD is highly promising, it is currently in the preclinical stage, having been conducted in animal models. Before these drugs can be considered for human treatment, extensive clinical trials are necessary. These trials will evaluate safety, optimal dosing, and efficacy in human patients. This process typically takes several years, so widespread availability for MASLD treatment is still some time away, pending successful human clinical validation.
A Glimmer of Hope for Liver Health
The growing understanding of MASLD’s mechanisms and the innovative approaches to treatment, particularly through drug repurposing, offer a significant glimmer of hope for millions worldwide. The University of Barcelona’s research on pemafibrate and telmisartan represents a vital step forward, showcasing a potential path to not only reverse fatty liver accumulation but also mitigate associated cardiovascular risks. As scientific efforts continue, focusing on rigorous clinical trials and exploring diverse therapeutic avenues, the future for MASLD treatment appears brighter, promising more effective and safer options for improving liver health and overall well-being.